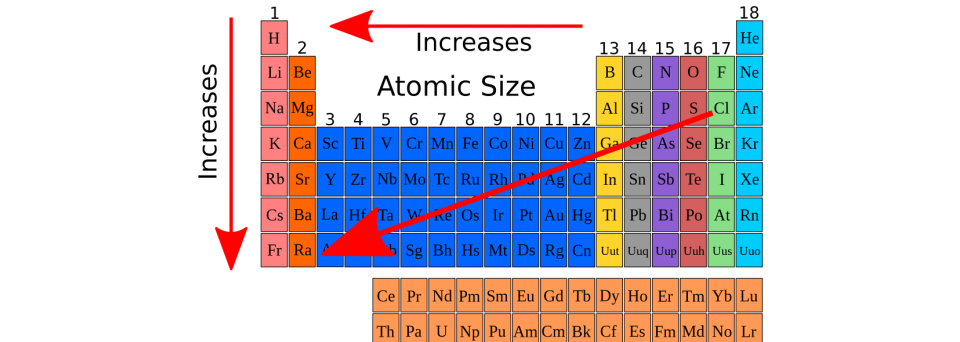
Hence the decrease in the radius is not as much as expected. This slightly offsets the increase in the positive charge in the nucleus. In these elements, from left to right, the differentiating electron enters into the inner d-orbitals that results in the reduction of nuclear attraction over the outermost electron. It is due to shielding effect of inner d-electrons. In the transition elements, the observed decrement in atomic size is very small across the period. Yet, it is misleading that the sizes of zero group elements are greater than those of corresponding halogens, since the only reported radii for noble gases are van der Waal's radii. This outweighs the increase in nuclear charge.Į.g., The order of atomic radius in IA group is Li Be > B > C > N > O > F < Ne Thus in groups, the atomic size increases down the group as the differentiating electron enters into the new quantum shell. However, a rough comparison of reported atomic radii is helpful in understanding the trends in the periodic properties of elements. Though it is strictly not possible to compare the radii of atoms in a given group or period since for each element the accurate atomic radii are not available and the values depend on the type of radii reported and the state of the substance as well as the nature of bonding interactions. Usually van der Wall's radii are 40% greater than crystal or covalent radii. It is especially applicable to noble gas elements. The half of inter nuclear distance between two closest atoms of different molecules attracted by van der Waal's forces. This is applicable to atoms in covalent substances. The half of inter nuclear distance between two atoms held together by a covalent bond. Since the atoms in a metallic crystal are closely packed, the crystal radius is considered to be more accurate radius of atom. The half of inter nuclear distance between two adjacent atoms in a metallic crystal is known as crystal radius or metallic radius or most of the time as atomic radius. The atomic radii are broadly divided into three types as shown below depending on the nature of substance and bonding in it etc. Hence we take the atomic radius as the average distance. The exact position of an electron is uncertain and cannot be determined accurately. The average distance between the center of the nucleus and the electron cloud of outermost orbit is called atomic radius.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
